I mentioned at the end of A Clinical Trial – Patient Journey – Part I that I had originally planned not to share my experience until after the placebo arm was over. On reflection, I realised that would in fact be unintentionally biased reporting, because events during, and the experience of, that first six months, when I may or may not be on the active intervention, are valid when discussing clinical trials from a patient perspective.
I had convinced myself I would get the placebo. I do not have a history of winning at chance! The lottery? Yeah, nah. Raffles? Not a hope. Scratchies (are they still a thing)? Never! I did once win $100 in a work event of some sort, but that is hardly indicative of a great winning streak. Psychologically, it is easier to plan on having a further toughish six months than be disappointed if nothing happens. Makes sense, I think!
In Part I, I also mentioned I thought I may get an early indication based on any skin improvement. This graph is from psoriasis clinical trials.

As can be seen from the above, results in the skin trials were seen very quickly. Although the trial in which I am participating is for the arthritic condition, the skin condition will also be treated.
Let’s pick up where we left off, at the baseline visit.
Baseline
After dealing with all the paperwork, doctor consultation, having bloods and urine taken, temperature and blood pressure checked, joint and enthesitis assessment and answering a lengthy quality of life type questionnaire on an iPad, it was time for the injections. I remember watching it go in (yes, I watch injections, apologies to squeamish readers) and thinking, “There’s my saline solution dose for the day”. One into the abdomen, one into the thigh.
From November 2018, I had been having UV B light therapy for the psoriasis. It really works well, but one of the criteria of the trial is no light therapy. Makes sense – the aim is to test the efficacy of the drug and that would be most difficult if certain other therapies (see explanatory comment below) were being employed at the same time. So I had to stop light therapy a specific period of time prior to the baseline visit.
The following two photos I took July 6. While with UV B and methotrexate it had reduced dramatically, once off both those therapies the skin had flared again. My baseline visit was July 17. I’d stopped the UV B therapy a little earlier than I otherwise would have, because I’d stopped in preparation for one trial, then ended up on the sister trial with a later start date.
These photos are embarrassing to share, nor are they anything like “clinical quality” as I just snapped them with my phone, but they are more meaningful that quoting percentages. If my articles are to be useful to readers, I have to be prepared to drop the mask of normality so many of us wear a lot of the time. I wanted my own baseline for comparison. We forget how things looked, over time: I wanted to be able to look at my skin further down the line on the trial and be able to see (or not see) any change. As this trial is for psoriatic arthritis, not psoriasis, no photos are part of the trial documentation.
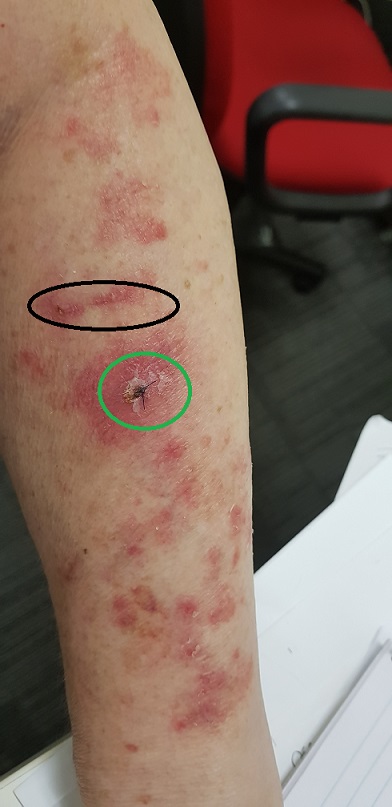
The image on the left is a patch on my leg. The image on the right is my upper chest, just below the collar bone. You can see older images of my arm on “I am Medication Free! For Now, Anyway…”

The skin makes swimming and hydrotherapy embarrassing. Rightly or wrongly, I worry other swimmers are going to think I’m contagious. It just looks…..horrid. Worth mentioning, perhaps, this sort of skin involvement is new to me – when I say new, I mean I went through MANY years of my life with the odd tiny patch which would disappear, or absolutely none. I am not used to this.
I’ve shared this swollen foot photo before, this was February, just as DMARD number two washed out of my system. This is the psoriatic arthritis aspect. The right second toe is clearly also swollen – dactylitis. Yes, you may indeed compliment me on my choice of polish!

The joint and enthesitis assessment is quite lengthy because it involves an independent assessor testing the many finger and toe joints as well as, jaw (TMJ), feet, elbows, shoulders, sternum, knees and hips (SIJ). It seems as if, if you have a joint, they will assess it! I never think of my toes being sore, aside from the big toes, yet it is surprising just how sore they are when a relatively light test pressure is applied. I knew my thumbs were sore, but all those toe joints: ouch, ouch, ouch. My fingers, aside from the thumbs, are OK. Even the thumbs are not bad – the soreness doesn’t stop me doing anything AT THIS TIME: however my concern is disease progression. I want, if at all possible, to stop progression.
The assessor also looks at and records the percentage of the body showing skin involvement.
At the point of the above photo, I could not fit my runners on at all – that makes exercise difficult as the gym frown on bare feet for safety reasons. The swelling had reduced a bit by July, but I was still struggling to wear runners for any length of time. The assessor, using a clinical approach rather than my layperson’s visual assessment, flagged many of my toe joints as swollen as well as sore. We get used to looking at our own bits and pieces and over time the abnormal can start to look normal.
Which injections you get is controlled by a mysterious person far, far away. To that mysterious person I am just a number. My co-ordinator emails the mysterious person, who I suspect at baseline tosses a coin to decide which arm I’m in: active or placebo. Of course I am joking – it is likely much more scientific than that. Now I’m curious – I must ask next visit!
An email comes back advising which two numbered boxes are to be used for my participant number. I wanted to keep the boxes as souvenirs (main photo) but they have to be kept for the trial auditors to check I was given what I was supposed to be given.
None of us, the investigator, or my co-ordinator, or I know what is in those syringes. It is a 50/50 lottery at baseline.
After the injections the trial participant (i.e. me) sits around for two hours in case of an adverse reaction.
Take a book or iPad or Kindle with you is my advice! You may have another participant, of your trial or another trial, to chat to or you may be sitting alone. Very comfy chairs, nice throw rug for colder days, but essentially a pretty boring two hours.
Then time to go home.
Please, PLEASE bear in mind that even if you were on the exact same trial as I am on, you may have a completely different experience to my experience. Your disease would not be at the exact same stage as mine, you may have more or less joints affected, you may have more or less skin involvement, you may suffer more or less fatigue than I do. So many variables. What I write is my specific experience. Your experience may be similar, or not.
The next stage in the loading dose. This happens four weeks after the baseline injections.
I know, I know, you want to know where I am on that graph! Can I fit my runners on now? Well, I gave the runner answer away in a recent article if you are paying attention. Be patient, all will be revealed in good time. I had to wait and see!
Explanation re Concurrent Therapies
Although light therapy is not allowed, participants may stay on up to two DMARDs while on the trial. After all, if the participant is in the placebo arm, if no concurrent therapies were allowed, those participants would effectively be receiving no treatment at all for six months. Each clinical trial will have restrictions specific to that trial.
I am also restricted in what other medications I can take, such as pain medications. Given I rarely take any pain meds (exercise is my pain management), that didn’t worry me. I can’t change my dietary supplements, although I can stay on the ones I started on e.g. Vitamin D as ordered by my endocrinologist, fish oil as recommended by my sports physician. There are rules around surgery during the trial.
These are all considerations when deciding whether to participate in a clinical trial.